
- Company overview The heart of SMC Vision & Philsophy Partnership Certifications Company culture
- Our service Design and Engineering Maintenance and Service Examine Production Line Upgrade and Transformation Storage and Logistics Processing, Trading and Distributor
- Management Our history Global responsibility Info Center
- Procurement center Internship
- Metal Steel Products Stainless Steel Products Aluminum Products Copper Products Galvanized Steel and PPGI Special Alloy Building Material
- Containers ISO Standard Container Equipment Container Storage Container Refrigerated/Reefer Container Offshore Container Container House Tank Container Container Fittings Container Trailer
- Gas Cylinder & Fire Extinguisher Cryogenic Liquid Cylinder Oxygen Gas Cylinder Storage Tank CNG Gas Cylinder LPG Gas Cylinder Hydrogen Gas Cylinder Nitrogen Gas Cylinder Industry Gas Cylinder Fire Extinguisher
- Metal Machinery Forming Machine Cutting Machine Processing Machine Bending Machine Block Machine Other Machinery Motor Spare Parts
- Mechanical Products Miscellany Mooring Equipment Marine Equipment Vehicle Industry Pressure Vessel Conveyor Belt Laser Equipment Bearing
- Electrical System Power Distribution Automation Electrical Cable Solar Power System Electric Protection System Transformer Production Line Lighting System
- Project Plastic Pipes and Pipe Fittings Fiberglass Reinforced Plastic Pontoon System
Metals Classifications and Properties
Metals Classifications and Properties
Metals are the materials that make up most of the products in our daily lives from our phones, our homes, our cars, our keys, etc. The definition of a metal is a material that is typically opaque, shiny, and has good electrical and thermal conductivity. Metals themselves are defined and categorized based on their properties. Metals make up ninety-one of one-hundred eighteen elements on the periodic table of elements. But how much do you really know about metals, how they are classified, and their properties?
Properties of Metals
The chemical properties of metals vary based on the metal. Generally, metals are inclined to form cations through electron loss, reacting with oxygen in the air to form oxides over different timescales. Some metals may only take seconds to rust others take years. In addition to high density, physically metals have high electrical and thermal conductivity. Metals are malleable and ductile meaning they deform and can be shaped under stress without cleaving or cracking. They also are fusible, meaning that they can be melted or “fused” together. Metals are typically shiny and lustrous.
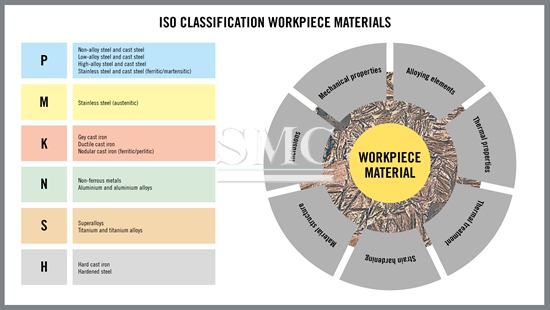
Categories of Metals
Metals are classified in a variety of ways. Base metals are metals that are easily oxidized or corroded and react easily with hydrochloric acid to for hydrogen. Some examples of base metals are nickel, lead, and zinc. These metals are generally inexpensive in comparison to precious metals such as gold and silver. Ferrous metals are metals that contain iron. In fact, ferrous is derived from the Latin word meaning “containing iron”. This does not mean it is pure iron; it can be any iron alloy such as steel. Often time’s ferrous metals are magnetic in nature, but not always. Noble metals are those metals that resist corrosion and oxidation. A majority of precious metals are classified in this category. Metals like gold, platinum, silver, rhodium, and palladium are all noble metals. These metals often have a high economic value due to their rarity and properties. Heavy metals are relatively dense metals or metalloids. Heavy metals often have niche uses and can be toxic. Most metal applications are made up of alloys. Alloys are mixture of two or more elements in which the main component is a metal. This is done to improve strength and reduce chemical reactiveness of certain metals. The most common alloys are stainless steel, carbon steels, and bronze.
Shanghai Metal Corporation is a trusted aluminum alloy, aluminum foil price, stainless steel price and stainless steel manufacturer, kinds of stainless steel in china.
