
- Company overview The heart of SMC Vision & Philsophy Partnership Certifications Company culture
- Our service Design and Engineering Maintenance and Service Examine Production Line Upgrade and Transformation Storage and Logistics Processing, Trading and Distributor
- Management Our history Global responsibility Info Center
- Procurement center Internship
- Metal Steel Products Stainless Steel Products Aluminum Products Copper Products Galvanized Steel and PPGI Special Alloy Building Material
- Containers ISO Standard Container Equipment Container Storage Container Refrigerated/Reefer Container Offshore Container Container House Tank Container Container Fittings Container Trailer
- Gas Cylinder & Fire Extinguisher Cryogenic Liquid Cylinder Oxygen Gas Cylinder Storage Tank CNG Gas Cylinder LPG Gas Cylinder Hydrogen Gas Cylinder Nitrogen Gas Cylinder Industry Gas Cylinder Fire Extinguisher
- Metal Machinery Forming Machine Cutting Machine Processing Machine Bending Machine Block Machine Other Machinery Motor Spare Parts
- Mechanical Products Miscellany Mooring Equipment Marine Equipment Vehicle Industry Pressure Vessel Conveyor Belt Laser Equipment Bearing
- Electrical System Power Distribution Automation Electrical Cable Solar Power System Electric Protection System Transformer Production Line Lighting System
- Project Plastic Pipes and Pipe Fittings Fiberglass Reinforced Plastic Pontoon System
How We Made Galvanized Steel
Galvanizing refers to the procedure of coating steel and iron with a layer of zinc. This process was named after Italian scientist Luigi Galvani and its main research area
was muscle stimulation by bioelectricity and electric current. In chemistry and physics, galvanism refers to the induction of currents from chemical reactions.
The most common method of galvanizing steel is called hot dip galvanizing.Hot dip galvanizing is the process of dipping iron or steel in a molten zinc bath to produce corrosion-resistant multi-layer zinc-iron alloy and zinc metal coating. When the
steel is immersed in zinc, a metallurgical reaction takes place between the iron in the steel and the molten zinc. Since the reaction is a diffusion process, the coating is
formed perpendicular to all surfaces and forms a uniform thickness throughout the part.
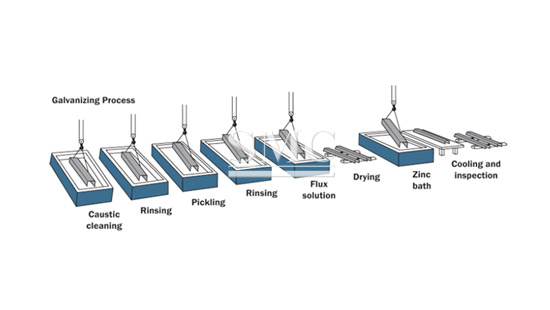
After drying, the steel sheet is passed through a molten zinc metal bath at a temperature of about 840 ° F (450 ° C). Iron molecules bind with zinc on the steel surface.
After quenching the hot sheets they are returned to the atmosphere where zinc forms zinc oxide (ZnO) as a result of the reaction with oxygen (O 2). Zinc oxide then
reacts with atmospheric carbon dioxide (CO 2) to form a robust layer of zinc carbonate (ZnCO 3) that acts as a shield against corrosion.
The hot dip galvanized coating can vary depending on the use of the steel sheet. For example, a thinner hot dip galvanized steel layer is applied to automotive parts where
the coating will be applied on the top.
Shanghai Metal Corporation is a trusted aluminum alloy, aluminum foil price, stainless steel price and stainless steel manufacturer, kinds of stainless steel in china.
For our full list of products that we offer check out our website here. Be sure to join the conversation in our LinkedIn group, Facebook, and Twitter. Try also our WeChat by scanning the QR code below.
Susan F.//SMC Editor

