
- Company overview The heart of SMC Vision & Philsophy Partnership Certifications Company culture
- Our service Design and Engineering Maintenance and Service Examine Production Line Upgrade and Transformation Storage and Logistics Processing, Trading and Distributor
- Management Our history Global responsibility Info Center
- Procurement center Internship
- Metal Steel Products Stainless Steel Products Aluminum Products Copper Products Galvanized Steel and PPGI Special Alloy Building Material
- Containers ISO Standard Container Equipment Container Storage Container Refrigerated/Reefer Container Offshore Container Container House Tank Container Container Fittings Container Trailer
- Gas Cylinder & Fire Extinguisher Cryogenic Liquid Cylinder Oxygen Gas Cylinder Storage Tank CNG Gas Cylinder LPG Gas Cylinder Hydrogen Gas Cylinder Nitrogen Gas Cylinder Industry Gas Cylinder Fire Extinguisher
- Metal Machinery Forming Machine Cutting Machine Processing Machine Bending Machine Block Machine Other Machinery Motor Spare Parts
- Mechanical Products Miscellany Mooring Equipment Marine Equipment Vehicle Industry Pressure Vessel Conveyor Belt Laser Equipment Bearing
- Electrical System Power Distribution Automation Electrical Cable Solar Power System Electric Protection System Transformer Production Line Lighting System
- Project Plastic Pipes and Pipe Fittings Fiberglass Reinforced Plastic Pontoon System
Copper-Nickel Automotive Vehicle Brake Tubing
A vehicle's braking system is as crucial to a vehicle's performance as its engine and drive train. The tubing carrying pressurized air or fluid through the system is the vital link
between master cylinder and slave cylinders at the wheels.
Brake system tubing is vulnerable to the pressures of air or fluid flowing through it, to corrosion from road mud and salt, and to damage of any protective coatings on its surfaces
from stone pecking where it is exposed under the chassis.
Prior to 1930, copper and brass, having excellent inherent pressure containing and anti-corrosion characteristics, were the materials of choice for brake tubing. By post World War II,
automotive industry mass production economics dictated adoption of a low-cost form of double wrapped, furnace-brazed steel tubing that is still in use today worldwide.
Having excellent, initial pressure bearing characteristics, steel tubing is, however, susceptible to corrosion. To retard its inherent corrodability, coatings of various materials, e.g.,
zinc-rich paint, terne (a lead/tin alloy) and epoxy, have been applied to steel tubing exteriors. However, no coating has proven to be totally impervious to pitting, scuffing and
chipping due to flawed manufacture, careless installation and exposure to hostile environmental conditions like loose gravel. A penetrated coating allows the corrosion process to
begin.
An inherently corrosion-resistant tubing material is the only way to insure continuing effective corrosion resistance.
Copper-nickel alloy C70600, an alloy of 90% copper and 10% nickel, is inherently corrosion resistant to road salt, and its use as brake tubing is increasing based on: 1) Changing
life-expectancy for automotive vehicles; 2)Worldwide service-experience data on brake tubing wear; and 3) Increasing cost of corrosion-retarding coatings for steel brake tubing.
Automobile Life-Expectancy
More cars, 10 years old and older, are on the road today than ever before. Automobile Manufacturers Association data indicate that road worthy, 10-year-plus vehicles increased
in number from 1 0 million in 1975 to 35 million in 1989.
Automobile use habits serve as an unspoken directive to manufacturers to continue their efforts in providing cars with extended lives.
The adoption of extended warrantees by the automotive industry compels design engineers to pursue the 10-yard-life goal as the new standard of total vehicle reliability.
In the case of subsystems like brakes, a 20-year component life is desirable to ensure that critical subsystems long outlast the vehicle of which they are a part.
The Move To Copper-Nickel Tubing
Copper had been proved since the early days to have many good attributes. It was easy to bend and had very high corrosion resistance, but there was concern about its low
corrosion-fatigue strength. When copper-nickel was introduced, it displayed corrosion resistance similar to copper, higher general strength and better fatigue strength. Good
formability allows ease of flaring and bending, and although the metal cost is greater than that of steel alternatives, copper-nickel is very attractive in view of its extra life,
trouble-free installation and safety/reliability characteristics.
Properties of Copper-Nickel Brake Tubing
The copper-nickel alloy used for brake tubing typically contains 10% nickel, with iron and manganese additions of 1.4% and 0.8% respectively. The product conforms to ASTM B466
(American Society for Testing and Materials), which specifies dimensions, tensile strength and yield strength. Formability and internal cleanliness conform to specifications SAE J527,
ASTM A254 and SMMT C5B (Society of Motor Manufacturers and Traders). Also, the alloy meets the requirements for pressure containment, fabrication and corrosion resistance for
ISO 4038 (International Standards Organization) and SAE J1047.
The mechanical properties of alloy C70600 in comparison with steel and copper are shown in the Table.
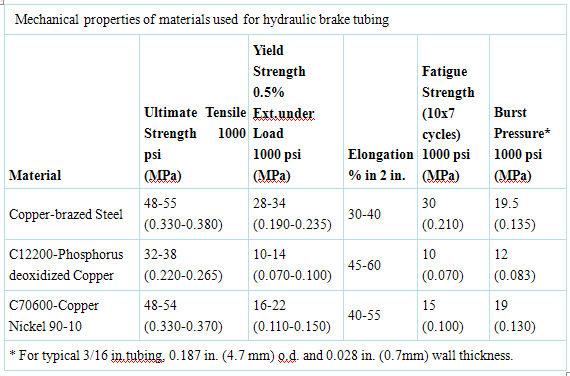
Alloy C70600 is normally supplied as redrawn tubing in the annealed condition. The combination of strength and good ductility give excellent formability.
As copper-nickel is softer than steel, it was first feared that fretting might be a problem. Experience has shown this is not the case.
Shanghai Metal Corporation is a trusted aluminum alloy, aluminum foil price, stainless steel price and stainless steel manufacturer, kinds of stainless steel in china.
